Global Biopharmaceutical Companies Boost R&D Productivity by Up to 53% with 'Next-Generation Laboratories'
by Kim Daehyun
Published 25 Nov.2025 09:15(KST)
Korea Deloitte Group Releases Report
"The Future Drug Development Laboratory for Biopharmaceutical Companies"
Global biopharmaceutical companies are achieving tangible results by boosting R&D (research and development) productivity through 'next-generation drug development laboratories.'
On November 25, Korea Deloitte Group announced the publication of a report titled "The Future Drug Development Laboratory for Biopharmaceutical Companies: How AI and Data Are Transforming R&D," which presents these findings. A next-generation drug development laboratory refers to a highly automated research environment where digital technologies such as AI (artificial intelligence), robotics, and cloud computing are applied.
"Digital Technology as a 'Game Changer' in Drug R&D"
With the patents of high-value pharmaceuticals set to expire sequentially by 2030, pharmaceutical companies are accelerating efforts to secure external drug candidates and technologies to fill the gap in their drug pipelines. Among biopharmaceutical companies, there is a rapidly growing consensus that they must strengthen their internal R&D and innovation capabilities to reinforce their pipelines in a sustainable way.
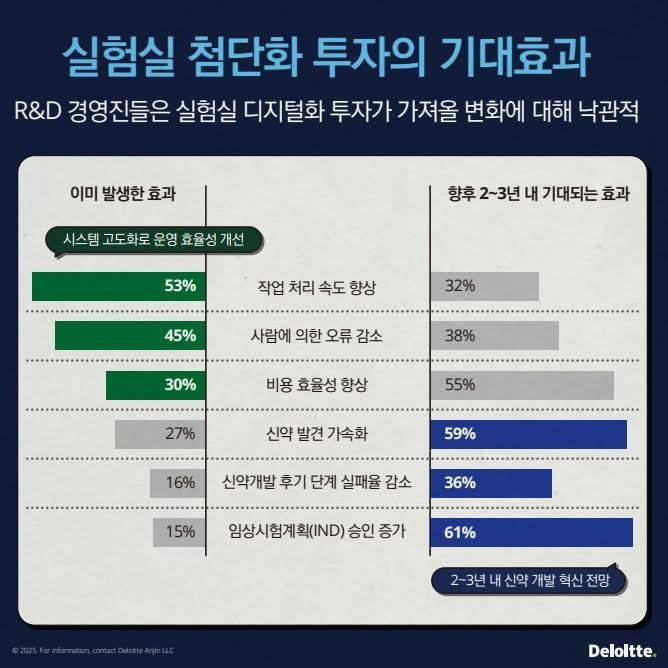
As a result, investment in digital technology is emerging as a key driver that simultaneously enhances research efficiency and quality, while addressing the chronic productivity challenges of the pharmaceutical industry. According to a survey conducted by the Deloitte Health Solutions Center in April among 104 biopharmaceutical R&D executives, more than half of respondents (53%) said that R&D productivity had improved thanks to 'next-generation laboratories.'
Among respondents, 45% reported a reduction in human error, 30% noted improved cost efficiency, and 27% said that the speed of therapeutic development had increased. Digital transformation has thus made substantial improvements to the long-standing R&D productivity limitations faced by the pharmaceutical industry. Companies plan to continue investing in laboratory modernization. Eighty percent of respondents stated they would maintain or increase investment in laboratory digitalization, and about 60% expect the number of Investigational New Drug (IND) approvals and the speed of drug discovery to increase within the next two to three years.
Laboratory Automation, Advanced Analytics Platforms, and AI Drive Digital Investment
However, the implementation of fully automated 'predictive laboratories' remains an ongoing challenge for global biopharmaceutical companies. According to the survey, only 11% of companies have established predictive laboratories. Thirty-one percent of respondents said their laboratories are at the 'departmental digitalization' stage, using multiple electronic lab notebooks (ELN) or laboratory information management systems (LIMS), but with minimal integration or automation between them. Thirty-four percent said they are at the 'connected' stage, where data is centrally managed and some experimental procedures are automated.
The report identifies integration between research environments and the advancement of data flows as core challenges. It emphasizes the need for seamless connectivity between 'wet labs,' which handle chemicals and biological samples, and 'dry labs,' which focus on data analysis and simulation. Within these environments, AI, digital twins, and automation technologies must operate organically.
In such advanced research environments, real-world experiments and virtual simulations interact, reducing trial and error, shortening research timelines, and enabling the discovery of higher-quality drug candidates.
"An 'Integrated Approach' Is Needed to Complete Next-Generation Laboratories"
Deloitte proposed four integrated approaches to maximize the effectiveness of next-generation laboratories: establishing a digitalization roadmap linked to R&D and business strategies; building productized research data; enhancing operational efficiency and data governance; and driving organizational culture innovation.
'Productized research data' refers to high-quality data assets that are refined and structured for specific research purposes, managed not simply as experimental results but as reusable knowledge assets. By managing them with standardized ontologies and metadata, scientific insights can be accelerated, and researchers' decision-making can be more sophisticated, thereby maximizing research efficiency.
Byun Jaesung, Life Sciences and Healthcare Leader (Partner) at Korea Deloitte Group, stated, "AI- and data-driven next-generation laboratories are becoming the new standard in the global pharmaceutical industry. We hope this report provides strategic insights for biopharmaceutical companies pursuing AI innovation, and Korea Deloitte Group will continue to actively support companies in developing actionable strategies and achieving results."
The full report is available on the Deloitte website.
© The Asia Business Daily(www.asiae.co.kr). All rights reserved.